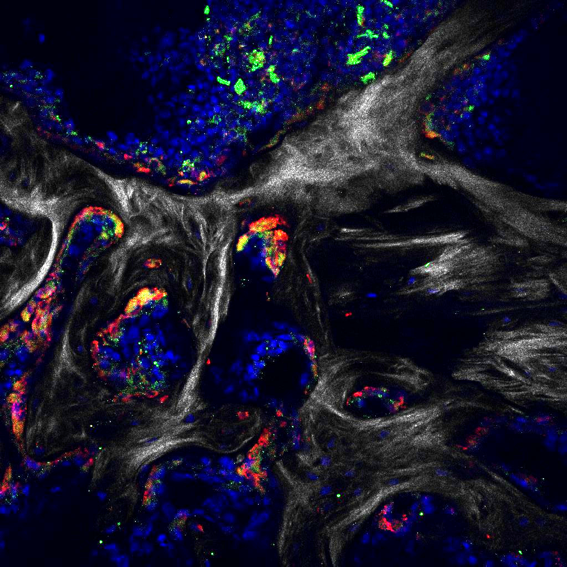
With an ever-increasing number of bone fractures, orthopedic surgeons are constantly faced with patients with delayed unions or non-unions. Aminocaproic acid has been approved and used in joint replacement and spinal fusion surgeries as an anti-fibrinolytic to limit perioperative blood loss. The efficacy and safety of aminocaproic acid has been studied extensively, and it has been shown to have a very safe and favorable risk profile. We are investigating aminocaproic acid as a pro-osteogenic agent and testing whether it can be used as an adjunct to enhance bone regeneration after long-bone fracture. This project is funded by the Orthopedic Trauma Association (OTA).
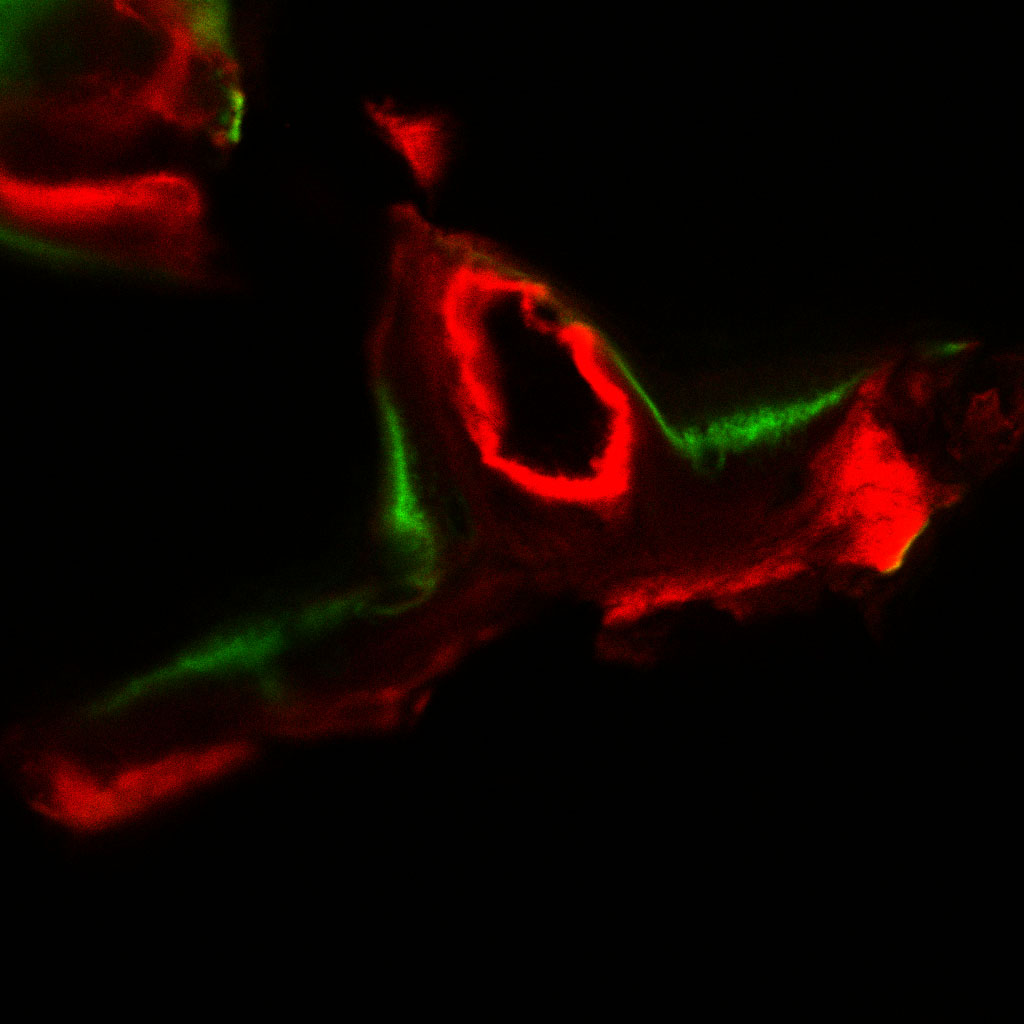
Adult fracture repair not only recapitulates embryonic bone development, it seems that bone cells maintain their embryonic ‘Postcode’ well into adulthood. Previous experiments have shown that stem cells transplanted from leg bones into fractured jaws failed to produce new bone (P. Leucht et al. Development 135, 2845–2854; 2008). Interestingly, the uncooperative stem cells continued to express a gene, Hoxa11, that acts as a kind of embryonic 'Zip code' for the leg. These findings have broad implications. They validate the concept of nonequivalence - that seemingly identical cells differ if they come from different places in the embryo. More pragmatically, if some stem cells have positional memory, surgeons may need to make sure that they take stem cells from the right location to heal damaged tissues. Here, we will further analyze the underlying molecular mechanism that confers neural crest derived osteoprogenitor cells with greater plasticity and therefore more regenerative potential.
This project is funded my the Musculoskeletal Transplant Foundation (MTF).
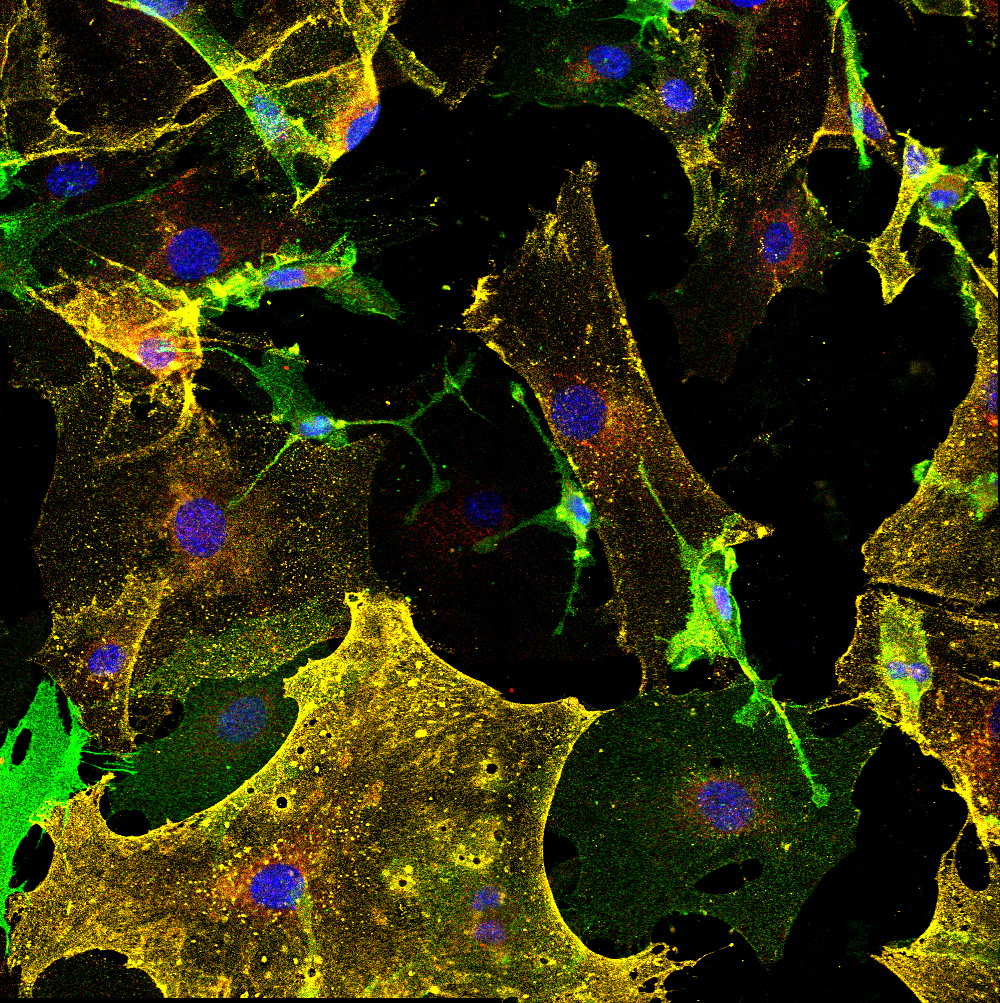
Notch and Wnt signaling are two pathways that govern bone regeneration and healing after injury. In this current study, funded by the NIH, we are investigating the intricate interplay of these two pathways during the early stages of bone regeneration.
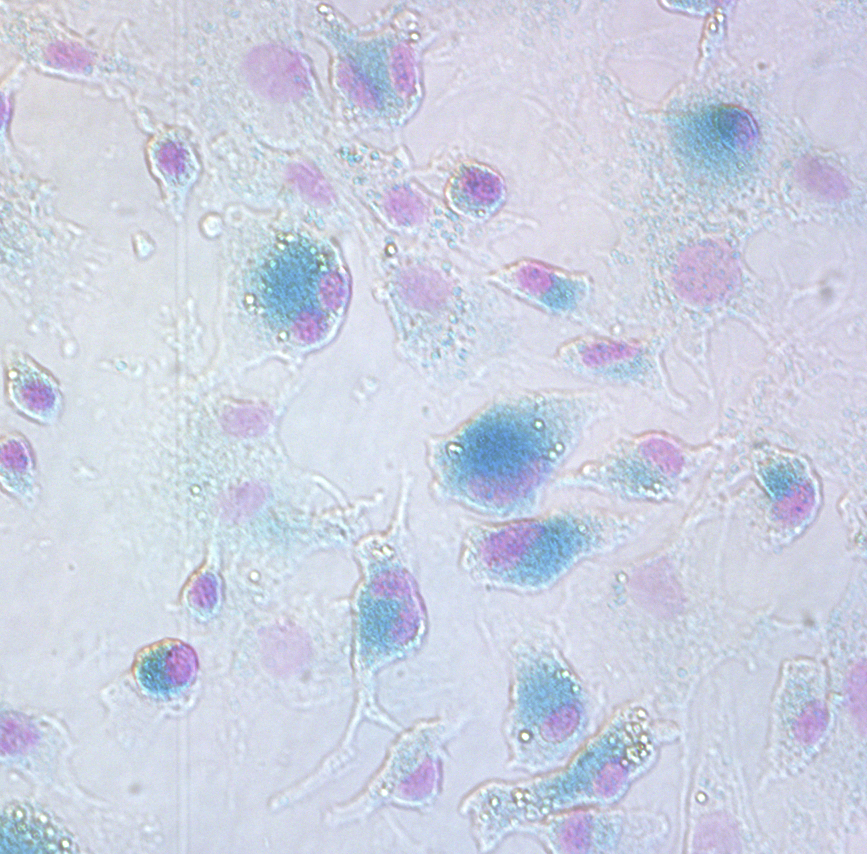
Aging has a dramatic effect on regeneration of all tissues, bone included. When a young adult fractures a bone, stem cells residing within the bone marrow cavity proliferate and differentiate into osteoblasts. This process results in regeneration of the skeletal element. In the elderly, however, this process is often inadequate, which will result in a delayed healing response. Chronic inflammation has recently been linked to a decline in regenerative potential in aged skin, skeletal muscle and nervous system. Accumulation of senescent cells in aging tissues has been suggested as the driver of chronic inflammation, as these cells secrete pro-inflammatory factors, which affect the entire surrounding cellular milieu, further stimulating inflammation. We are currently investigating the effects of age-related chronic inflammation, or inflammaging, on skeletal stem cell function and further if the observed negative effects are reversible.



